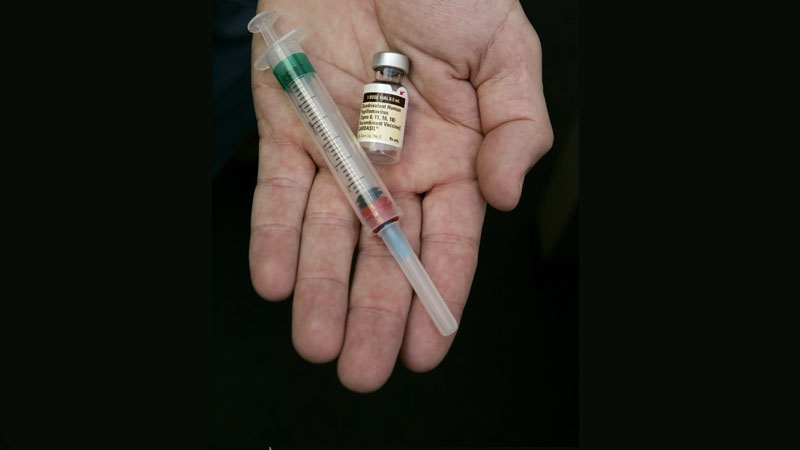
Health Canada has approved Gardasil 9, a second-generation HPV vaccine.
Gardasil 9 is designed to prevent cervical, vulvar, vaginal and anal cancers caused by nine HPV strains , five more than the original Gardasil vaccine which has been in use since 2006.
The earlier version covered HPV types 6, 11, 16, and 18. Gardasil-9 also covers types 31, 33, 45, 52, and 58 which researchers say are responsible for roughly one in five cases of cervical cancer.
The vaccine is also used to prevent genital warts caused by HPV types 6 and 11.
Gardasil 9 was approved for use in the United States in December and received the green light in Canada on February 5. It is recommended for girls and women aged 9 to 45 and boys/men aged 9 to 26. It is delivered in three doses given over six months and company officials say they expect it will be available before the end of this spring.
A Merck & Co Inc.-sponsored study in some 13,000 teens and girls, found Gardasil 9 was 97 per cent effective in preventing cancers caused by the five additional HPV types. Research also suggests the 9-strain vaccine can be safely given to those already vaccinated with the 4-strain product says Dr.Eduardo Franco, Director, of the Division of Cancer Epidemiology at McGill University, who has also served as an advisor to Merck.
Some doctors expect provinces to eventually switch from the 4-strain vaccine to the 9-strain product if Canadian vaccine-advisory groups recommend its use.
Injection site pain, swelling, redness, and headaches were the most commonly reported side effects. Fever, allergy to the vaccine, asthma attack, and tonsillitis were also noted as rare side effects. No vaccine related deaths were reported.
There are more than 100 types of human papillomavirus that can infect many parts of the body. Some types of HPV are sexually transmitted and can cause warts or other consequences such as cancer (e.g., cervical, penile and anal).
GlaxoSmithKline PLC sells an HPV vaccine called Cervarix, which targets two cancer-causing HPV strains.