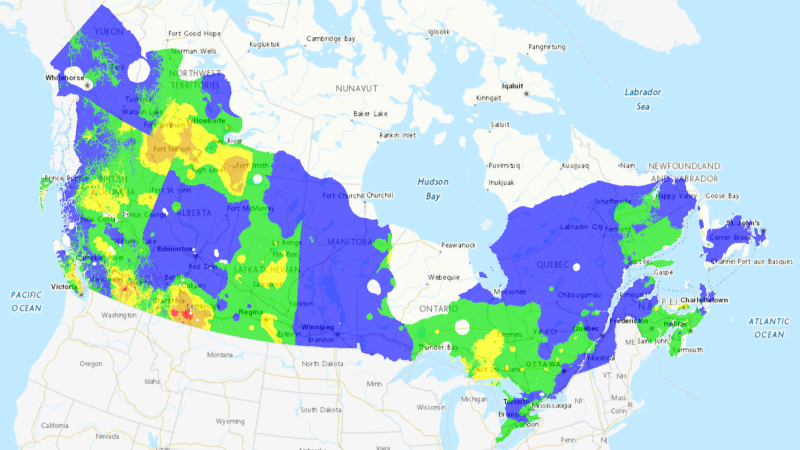
DEVELOPING Latest updates on the major wildfires burning in Canada
The 2024 wildfire season has begun, and it's shaping up to follow last year's unprecedented destruction in kind, with thousands of square kilometres already consumed.
A panel of U.S. health advisers on Tuesday narrowly backed a closely watched COVID-19 pill from Merck, setting the stage for a likely authorization of the first drug that Americans could take at home to treat the coronavirus.
A Food and Drug Administration panel voted 13-10 that the drug's benefits outweigh its risks, including potential birth defects if used during pregnancy.
The recommendation came after hours of debate about the drug's modest benefits and potential safety issues. Experts backing the treatment stressed that it should not be used by anyone who is pregnant and called on FDA to recommend extra precautions before the drug is prescribed, including pregnancy tests for women of child-bearing age.
The vote specifically backed the drug for adults with mild-to-moderate COVID-19 who face the greatest risks, including older people and those with conditions like obesity and asthma. Most experts also said the drug shouldn't be used in vaccinated patients, who weren't part of the study and haven't been shown to benefit.
The FDA isn't bound by the panel's recommendation and is expected to make its own decision before year's end. The pill is already authorized in the U.K.
The drug, molnupiravir, could provide a much-needed weapon against the virus as colder weather pushes case counts higher and U.S. officials brace for the arrival of the new omicron variant.
Merck hasn't specifically tested its drug against the new variant but said it should have some potency based on its effectiveness against other strains of coronavirus.
But that uncertainty frustrated many panelists as they grappled with whether to back the treatment for millions of Americans.
“With no data saying it works with new variants I really think we need to be careful about saying that this is the way to go,” said Dr. David Hardy of Charles Drew University School of Medicine and Science, who ultimately voted to back the drug.
The panel's narrow-but-positive recommendation came despite new data from Merck that paint a less compelling picture of the drug's effectiveness than just a few weeks earlier.
Last week, Merck said final study results showed molnupiravir reduced hospitalization and death by 30% among adults infected with the coronavirus, when compared with adults taking a placebo. That effect was significantly less than the 50% reduction it first announced based on incomplete results.
That smaller-than-expected benefit amplified experts' concerns about the drug's toxicity for fetuses.
FDA scientists told the panelists earlier Tuesday that company studies in rats showed the drug caused toxicity and birth defects when given at very high doses. Taken together, FDA staffers concluded the data “suggest that molnupiravir may cause fetal harm when administered to pregnant individuals.”
FDA is weighing a blanket restriction against any use in pregnant women or allowing it in rare cases. Some panelists said the option should be left open for pregnant mothers who have high-risk COVID-19 and may have few other treatment options.
Dr. Janet Cragan, who backed the drug, said that even with tight restrictions some pregnant women would inevitably take the drug.
“I don't think you can ethically tell a woman with COVID-19 that she can't have the drug if she's decided that's what she needs,” a panel member and staffer with the Centers for Disease Control and Prevention. “I think the final decision has to come down to the individual woman and her provider.”
Merck's drug uses a novel approach to fight COVID-19: It inserts tiny errors into the coronavirus' genetic code to stop it from reproducing. That genetic effect has raised concerns that the drug could spur more virulent strains of the virus. But FDA regulators said Tuesday that risk is theoretical and seems unlikely.
While Merck and its partner Ridgeback Biotherapeutics were the first to submit their COVID-19 pill to the FDA, rival drugmaker Pfizer is close behind with its own pill under review.
Pfizer's drug is part of a decades-old family of antiviral pills known as protease inhibitors, a standard treatment for HIV and hepatitis C. They work differently than Merck's pill and haven't been linked to the kind of mutation concerns raised with Merck's drug.
Pfizer said this week that its drug shouldn't be affected by the omicron variant's mutations.
The U.S. government has agreed to purchase 10 million treatment courses of Pfizer's drug, if it's authorized. That's more than three times the government's purchase agreement with Merck for 3.1 million courses of molnupiravir.
Both drugs require patients to take multiple pills, twice a day for five days.
------
The Associated Press Health and Science Department receives support from the Howard Hughes Medical Institute's Department of Science Education. The AP is solely responsible for all content

The 2024 wildfire season has begun, and it's shaping up to follow last year's unprecedented destruction in kind, with thousands of square kilometres already consumed.
Veteran TSN broadcaster Darren 'Dutch' Dutchyshen, one of Canada’s best-known sports journalists, has died. He was 57. His family says 'he passed as he was surrounded by his closest loved ones.'
A ‘lifetime of abuse’ led Dallas Ly to snap and repeatedly stab his mother inside their Leslieville apartment in 2022 but he never intended to kill her, his defence lawyers argued during at his murder trial in Toronto on Thursday.
A burgeoning track star says his dream of going to the Olympics is being derailed by a deportation order after Immigration officials rejected his family’s claim for asylum
A father has been charged with second-degree murder in the stabbing death of his 34-year-old daughter in southern Quebec.
A medical examiner says a Massachusetts teen who participated in a spicy tortilla chip challenge died from ingesting a substance 'with a high capsaicin concentration.'
A Montreal father who kidnapped his daughter who has autism and lied to police when they asked where she was should serve three years in prison, a Crown prosecutor said.
The province’s health minister and solicitor general are urging Toronto to rescind its request to decriminalize simple possession of small amounts of drugs for personal use, calling the proposal 'misguided' and 'disastrous.'
Prime Minister Justin Trudeau says he has "issues" with the Progressive Conservative government of New Brunswick.

A Starbucks fan — whose name is Winter — is visiting Canada on a purposeful journey that began with a random idea at one of the coffee chain's stores in Texas.
Members of Piapot First Nation, students from the University of Winnipeg and various other professionals are learning new techniques that will hopefully be used for ground searches of potential unmarked grave sites in the future.
ALS patient Mathew Brown said he’s hopeful for future ALS patients after news this week of research at Western University of a potential cure for ALS.
When Adam Kirschner wrote 'Slap Shot,' he never imagined the song would be embraced by his favourite team.
A team is ready to help an entangled North Atlantic right whale in the Gulf of St. Lawrence.
A $200 reward is being offered by a North Vancouver family for the safe return of their beloved chicken, Snowflake.
Two daughters and a mother were reunited online 40 years later thanks to a DNA kit and a Zoom connection despite living on three separate continents and speaking different languages.
Mother's Day can be a difficult occasion for those who have lost or are estranged from their mom.
YES Theatre Young Company opened its acclaimed kids’ show, One Small Step, at Sudbury Theatre Centre on Saturday.