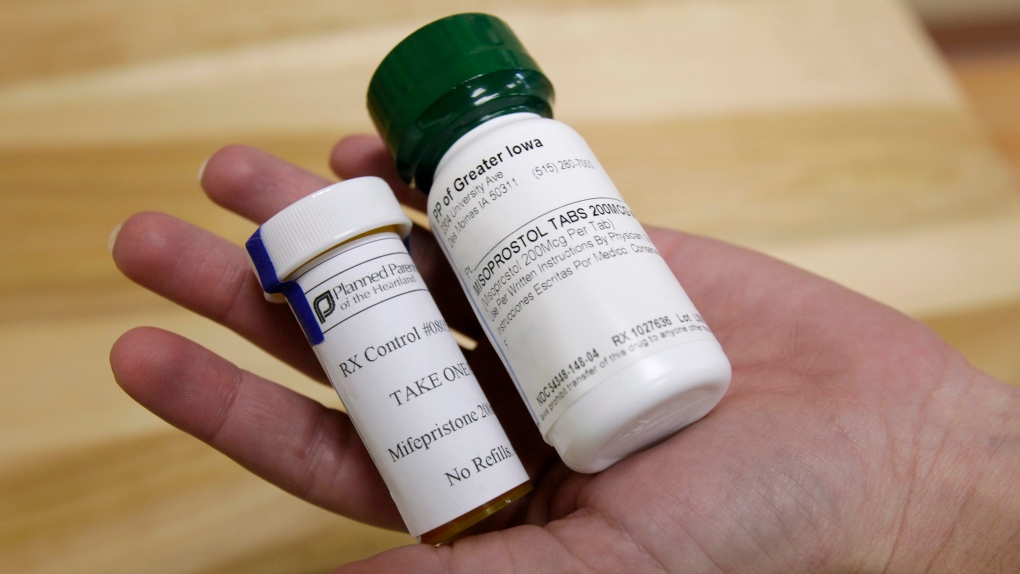
OTTAWA -- An organization that advocates for women's sexual health rights has given the provincial premiers a November deadline to provide universal access to Mifegymiso, a drug used to end a pregnancy.
Action Canada for Sexual Health and Rights is challenging the provinces to offer the medication free of charge to those who want it. New Brunswick, Quebec, Ontario and Alberta have already decided to offer it along with surgical abortions. Manitoba has also reportedly decided to offer medical abortions free of charge.
The deadline is the date of a fall meeting among provincial and territorial health ministers.
The abortion drug, also known as RU-486, hit the Canadian market last January, decades after it first became available in France. Health Canada authorized it for use terminating pregnancies up to seven weeks, while a 2016 article in the Journal of Obstetrics and Gynaecology Canada recommends its use up to 10 weeks.
But the cost -- up to $450 -- can be prohibitive to many women, advocates argue. At the same time, surgical abortions cost about twice what medical abortions do, but are covered under provincial health-care services.
"This is a question of avoiding a two-tiered system of abortion access in Canada," said Sandeep Prasad, Action Canada's executive director.
While Mifegymiso is covered for people on social assistance, and may be covered for those who have private benefits through their jobs, anyone caught in between would have to pay out-of-pocket for the medication.
"Those people will always then be choosing surgical abortion because medical abortion will not be a real option for them, and that's not a real choice," Prasad said.
"We need universal cost coverage."
Health Canada unclear on nurse practitioners prescribing
Prasad made the case Tuesday night in Edmonton at an event timed to coincide with the premiers' meeting, alongside other experts including Alberta Health Minister Sarah Hoffman and Barb Shellian, president of the Canadian Nurses Association.
Universal coverage for Mifegymiso is only one of the remaining barriers to access in Canada. While Health Canada has already had to clarify its guidelines for who can prescribe and dispense the drug, the Canadian Nurses Association would like to see a further clarification that nurse practitioners -- who are authorized to prescribe medication -- can issue prescriptions for Mifegymiso.
That ability is a pressing issue in communities that are too small or remote to have a doctor providing care. Canada has 5,000 nurse practitioners right now, of which "a large number ... provide primary care services in rural and remote Canada," Shellian said in an interview with CTVNews.ca.
"We don't believe that those women should be disadvantaged because they live in the northern part of our country, or that they are also perhaps in a correctional facility or they're being looked after by an agreement with the federal government, as some indigenous populations are," she said.
Shellian says the CNA has been working with Health Canada for nearly a year and calls it an error of omission.
"Nurse practitioners are prescribers of many drugs, including controlled substances, and they are expected to adhere to the stipulations that are put upon them by their college or their regulatory body," she said. "Nurse practitioners are quite able to prescribe Mifegymiso and ... provide the follow-up care that is identified by Health Canada."
The College of Nurses in Ontario has told its members that nurse practitioners can prescribe Mifegymiso.
Health Canada could update its guidelines to refer to authorized prescribers rather than physicians, Prasad noted.
"The promise of Mifegymiso is it's easy to roll out in rural and remote settings," he said.
"And so, if we have cost-coverage combined with a few other key interventions, such as ensuring that nurse practitioners are able to prescribe Mifegymiso, this will make a real difference to abortion access in this country."
The CNA also supports the drive for each province and territory to cover the cost of Mifegymiso.
A spokeswoman for Health Canada told CTVNews.ca that the product monograph it approved "is not a legally binding document," but a scientific summary of the information reviewed during the department's product assessment.
"Under the practice of medicine or pharmacy, which is regulated by provinces and territories, doctors, authorised prescribers such as nurse practitioners and pharmacists may administer or distribute drugs outside of approved condition of use to treat patients under their responsibility. Some jurisdictions have chosen to have access pathways that involve dispensing directly to patients by permitting doctors, nurse practitioners or pharmacists in those regions to distribute the drug according to those regional criteria," Rebecca Gilman wrote in an email.
Health Canada is currently reviewing the parent company's application to update the monograph, which has already been clarified twice. Once was to confirm patients do not have to take the drug in front of their physicians, and another confirmed that pharmacists -- not doctors -- can dispense it.