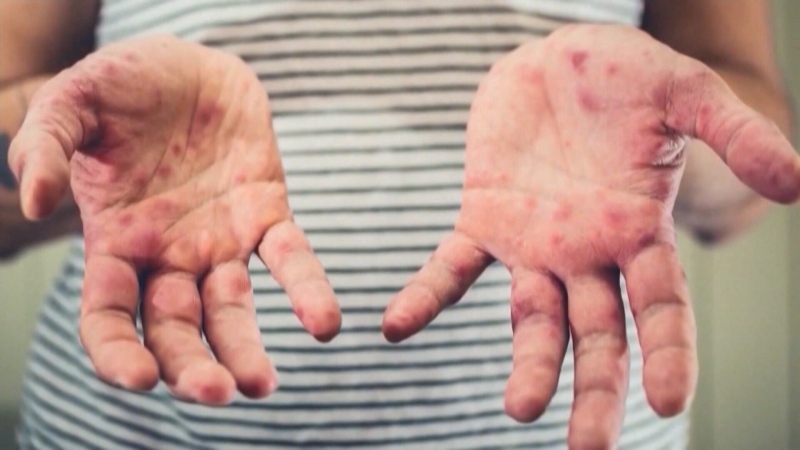
Lyme disease is a growing problem in Canada, with close to 1,500 cases reported to the Public Health Agency of Canada last year. With those numbers going up every year, many are asking why there isn’t a vaccine against the disease.
In fact, there was once was a vaccine on the market and it was highly effective. But mistaken concerns about the vaccine’s side effects soon spelled its demise, says Toronto-based physician-scientist Dr. Julielynn Wong.
“The vaccine company stopped making it for two reasons: low sales (Lyme disease was less common 20 years ago); and lawsuits,” she told CTV’s Your Morning Monday.
The vaccine was called LYMErix, and when it was approved for sale in the U.S. in December, 1998, sales were high. What made LYMErix unique was that it worked not by triggering antibodies in vaccine recipients, but by neutralizing the disease-causing bacteria within the tick itself.
When a tick would feed on a vaccinated human’s blood, the vaccine would enter the tick, and block the bacteria inside the tick‘s gut from entering the human.
The vaccine’s maker, SmithKline Beecham (currently known as GlaxoSmithKline), said its studies showed the vaccine was 78 per cent effective after three doses.
But within a year of the vaccine’s release, some users began to report they had developed neurological problems or arthritis shortly after vaccination.
The FDA began immediate studies to determine if the vaccine was responsible. They could find no link.
But by the time those studies were complete, patients who claimed to have been injured had already drummed up large support networks, and the resulting media coverage of their concerns led to drops in sales.
When vaccine users began to launch class-actions lawsuits, the vaccine maker decided they weren’t willing to spend the money needed to defend themselves in court. So the vaccine was pulled after just three years on the market.
Ever since, Lyme cases have continued to rise, and the ticks that carry it have spread to new areas where more people could become infected. That has many interested in reviving the vaccine, says Wong.
“Your dog can get a Lyme disease vaccine but you can’t,” notes Wong. “And a vaccine is really the only way to protect yourself from getting Lyme disease if you’re bitten by an infected tick.”
Now, a European vaccine maker, Valneva, is hoping to revive it. It has taken the same technology behind the LYMErix and developed its own vaccine, currently called VLA15, which would prevent six strains of Lyme.
The company says VLA15 is currently undergoing Phase 2 effectiveness testing and says it’s confident the vaccine doesn’t cause autoimmune reactions.
If all goes well, they say the vaccine could be ready to go to market in five years. That’s news that Dr. Wong say she would welcome because telling people to do tick checks does not appear to be working.
“Because the number of Lyme disease cases are going up, that tells us that tick prevention measures are not good enough to prevent Lyme disease cases,” she said.