MANILA, Philippines - The Philippine Department of Health said Friday it has put on hold its dengue immunization program after new findings by vaccine manufacturer Sanofi Pasteur that severe cases of dengue can occur in the longer term among those vaccinated without prior dengue infection.
Health Secretary Francisco Duque III said further recommendation will be released on Dec. 12 or 13 by the Strategic Advisory Group of Experts on immunization, an advisory body of the World Health Organization.
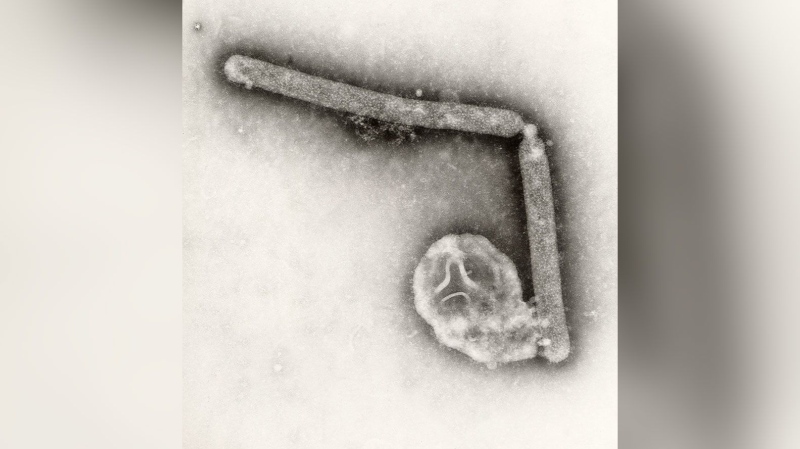
More than 730,000 public school children aged 9 and above in three highly endemic Philippine regions have received at least the first dose of Dengvaxia, the first licensed dengue vaccine that is manufactured by Sanofi. The Department of Health launched last year its 3.5 billion peso ($69.5 million) public dengue immunization drive - the world's first such program.
Sanofi said Wednesday that analysis based on up to six years of clinical data showed that in the longer term, more cases of severe dengue can occur following vaccination among people who have not had previous dengue infection. It said it will ask health authorities to update information provided to physicians and patients.
The analysis confirmed Dengvaxia provides persistent protective benefit in those who had prior infection, Sanofi said.
For those who were not previously infected by the dengue virus, the analysis found that vaccination prevented severe illness for at least 30 months, Duque told reporters.
He said there are currently no reports of severe dengue infection among those vaccinated. He did not say whether legal action will be taken pending conclusion of a review he has ordered of the contract and other documents on the Dengvaxia immunization.
Duque said the government will profile all those who received the vaccination and heighten its surveillance mechanisms. That will include mandatory history taking of those vaccinated, mandatory reporting of all hospitalized vaccine recipients regardless of symptoms and 5 years of post-vaccination surveillance.
Khristine Estrada-Cabanayan, a Sanofi spokeswoman in the Philippines, said the company is working closely with the Department of Health and will release a statement to address concerns regarding the vaccine.