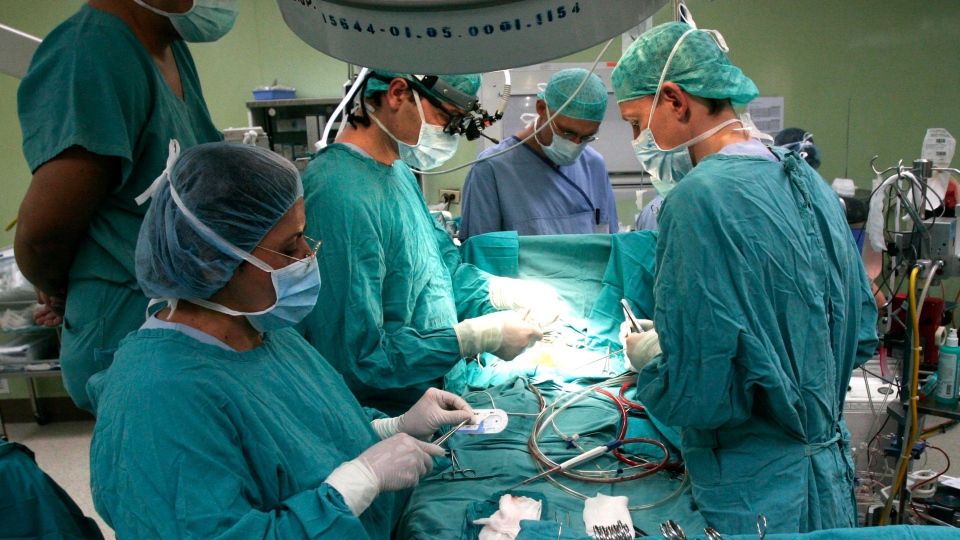
An alarming new study suggests that more than a third of heater-cooler devices, an instrument commonly used in open-heart surgeries, may contain water contaminated with deadly bacteria.
The research, presented at a U.S. conference by John Rihs, vice-president of laboratory services at Special Pathogens Laboratory, analyzed 89 heater-cooler units in 23 hospitals in Canada and the United States between 2015 and 2016.
A total of 33 units, or 37 per cent, tested positive for the bacteria M. chimaera.
Concerns about the bacteria’s presence have been raised in the past, but the new report offers a greater sense of how widespread the problem may be.
“The extent of contamination from such a rare organism in multiple units from all over the country was surprising," Rihs said in a press release issued Wednesday, as the research was presented at the annual conference of the Association for Professionals in Infection Control and Epidemiology (APIC).
Last fall, Health Canada and the U.S. Centers for Disease Control and Prevention (CDC) issued warnings about the risk of infection for anyone who underwent open-heart surgery since 2012. Public Health Ontario and the U.S. Federal Drug Administration have issued similar safety alerts.
Thousands of patients across Canada have since received letters warning them about the potential for infection and urging them to visit a hospital if they show certain symptoms.
Heater-cooler devices are rigged with specialized water tanks that help control a patient’s blood temperature during surgery. The water does not come into direct contact with the patient, but particles could enter their system if the water aerosolizes through the unit’s exhaust vent.
The devices are widely used in operating rooms across North America. Estimates suggest that about 60 per cent of heart bypass procedures performed in the U.S. use a brand of heater-cooler unit associated with the M. chimaera infections.
Identifying the infections can be particularly difficult. The nontuberculous mycobacteria grow slowly, so patients infected during surgery may not present with problems for up to five years after the procedure. Doctors say this means infections are likely under-reported.
Symptoms include fever, unexplained and persistent night sweats, unintentional weight loss, muscle aches, fatigue, as well as redness or pus along a surgical incision.
Diagnosis can be delayed for years or, in some cases, missed altogether.
In Canada, at least four patients have been diagnosed with the infections. In the U.S., 12 patients have died.
Richard Woodland from Lingan, N.S. suffered from an infection after undergoing open-heart surgery two years ago. He still doesn’t know whether his illness was the result of contaminated equipment.
“I’m very angry about it,” Woodland told CTV News. “The fact that I was sent home and not told about it, and I’m wondering how many more people might be out there.”
On a global scale, at least 100 heart surgery patients have been identified with M. chimaera infections after cardiac surgery, according to the CDC. The majority of infected patients had lengthy procedures that included implants, such as heart valves.
While the LiveNova/Sorin 3T heater-cooler device has been linked to the most infections, data from the FDA released in January reports that every similar device on the market has had some contamination reported, and that most devices have infected patients.
Health Canada issued an alert in February specifically raising concerns about the LivaNova/Stöckert 3T heater-cooler units.
All five manufacturers of heater-cooler units commercially available worldwide have had reports of contamination sent to the FDA through mandatory or voluntary reporting. Four of the five manufactures have had patient infections reported, and three have had associated deaths.
The research also identified four units colonized with Legionella, a bacterium that can lead to a flu-like infection and, in more serious cases, Legionnaires' disease. Several other strains of mycobacteria were also detected.
“Beyond M. chimera, we found other NTM species, Legionella, and fungi, indicating these units are capable of supporting a diverse microbial population," Rihs said.
The findings highlight the importance of closely monitoring cleaning and maintenance routines for heater-cooler devices, according to Linda Greene, president of the APIC conference.
"Hospitals must follow the cleaning and disinfection instructions provided in the manufacturer's device labeling, as well as updated communications from the FDA and CDC," Greene said.
Dr. Louis Perrault, the head of the department of surgery at the Montreal Heart Institute, says that if a device is found to contain the harmful bacteria, it should be taken out of use.
“I think contaminated units should most likely be set aside, replaced if possible,” Perrault said.
With a report from CTV’s medical specialist Avis Favaro and producer Elizabeth St. Philip