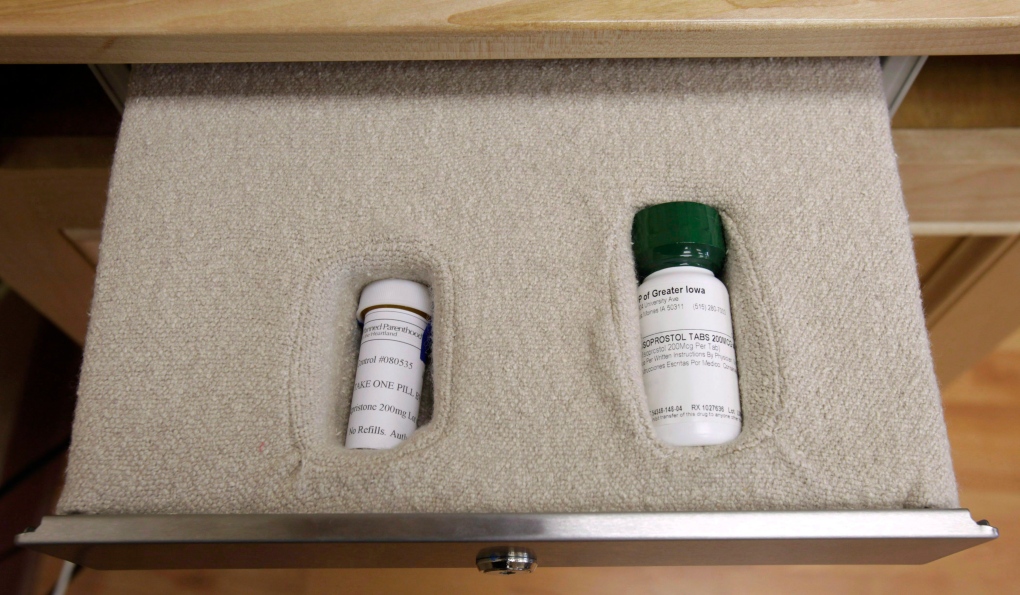
TORONTO -- The much-anticipated arrival of the abortion pill Mifegymiso in Canada has been delayed until the new year.
The drug's Canadian distributor, Celopharma Inc., says it now expects to launch "some time in January."
Company president Paula Tenenbaum gave few details in a terse email exchange but said it was related to "a change in manufacturing site."
Last month, Tenenbaum said Mifegymiso was on track for a November debut in Canada.
Health Canada approved the drug in July with several controversial restrictions, including that it only be dispensed by a doctor to a patient, which critics feared would restrict access for patients in rural and remote areas.
It's also limited to use no more than seven weeks after a woman's last period and comes with a steep $300 price tag.
The drug has been available elsewhere for nearly 30 years and is approved for use in more than 60 countries with varying restrictions.
Last month, Tenenbaum said Celopharma was working on a revised submission to Health Canada that would increase the recommended use to up to nine weeks after a woman's last menstrual period.
She also expressed her preference that pharmacists dispense the drug directly to patients.
Tenenbaum said Monday that she hoped to get a response from Health Canada "some time late December."
A spokesman for Health Canada said the federal agency does not disclose details about the status of drug approval applications, but "firmly supports a woman's right to choose."
"The department is committed to minimizing any undue delays in the review of any submission. Health Canada makes all drug approval decisions based on a detailed scientific review, and we continue to work with Celopharma on its application."
Mifegymiso is a combination of two drugs taken on separate days.
Mifepristone is taken first to block the hormone progesterone, which helps prepare the lining of the uterus for pregnancy. This causes the lining to break down and shed.
Then it's followed 24 to 48 hours later by misoprostol, which causes the uterus to contract and expel the pregnancy.